|
Now in this electron configuration of potassium, we have to see the total number of electrons present in the highest energy level. So from the Aufbau principle, we can get the electron configuration of the potassium atom as 1s 2 2s 2 2p 6 3s 2 3p 6 4s 1.According to the Aufbau principle, the electrons will be filled first in 1s orbital, then in 2s orbital, then in 2p orbital, and so on….Now we have to fill these 19 electrons in the atomic orbitals according to the Aufbau principle.The potassium atom has a total of 19 electrons because its atomic number is 19 and it is a neutral atom.To write the electron configuration of potassium, we should first know the total number of electrons present in a potassium atom.Now let’s try to find the electron configuration of Potassium by using the Aufbau principle.įollow the steps mentioned below to get the electron configuration of Potassium. Orbitals Maximum capacity of electrons s 2 p 6 d 10 f 14 Now there are many methods to write the electron configurations, but here I will show you the easiest method, i.e by using Aufbau principle.Īufbau principle: The Aufbau principle simply states that the orbitals with the lower energy are filled first and then the orbitals with higher energy levels are filled.Īccording to the Aufbau principle, the orbitals are filled in the following order:ġs, 2s, 2p, 3s, 3p, 4s, 3d, 4p, 5s, 4d, 5p, 6s, 4f, 5d, 6p, 7s, 5f, 6d, 7p, and so on.Īlso the maximum number of electrons that can be accommodated in s, p, d & f orbitals are mentioned in the below table. 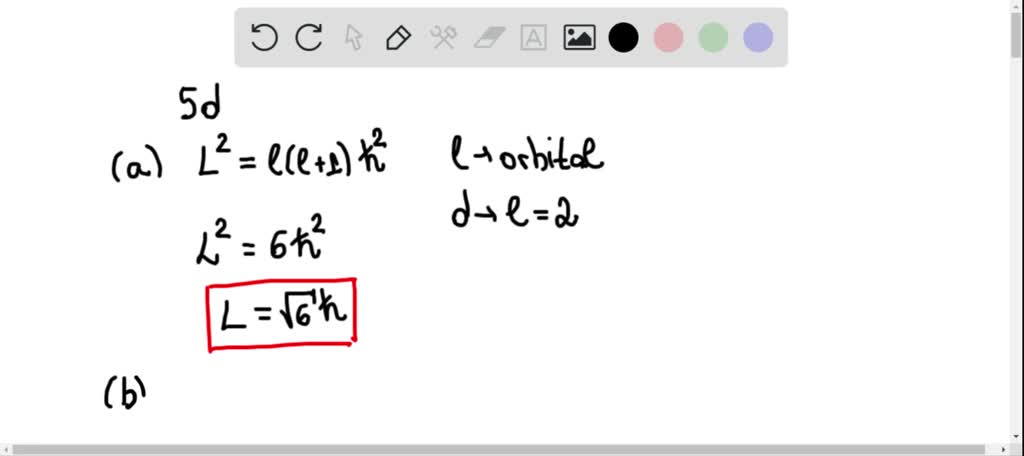
If you want to find the valence electrons of potassium from its electron configuration, then you should know its electron configuration first. Method 2: From the Electron Configuration Now let’s see another method for finding the number of valence electrons in potassium. In this way, by knowing the position of potassium element in periodic table, you can easily find its valence electrons. So, as the potassium element is present in group 1, it has 1 valence electron. More specifically, you have to see the group wise position of Potassium element in the periodic table.įrom the above image, you can see that Potassium (K) is present in the group 1 of periodic table. This citation format is based on MLA.To find out the valence electrons of Potassium, you have to see the position of potassium in the periodic table. Also replace URL for the actual url of this page (The stay, ok?). If we look at the electron configuration of potassium (K), we see that it has one electron: Ar4s 1. You are correct in that it has 8 valence electrons, not 0. Now replace dd, mmmm and yyyy with the day, month, and year you browsed this page. If we take that one valence electron away, it makes sense to say that it now has zero valence electrons because '1 - 1 0'. "Valence Electrons in Potassium (K) [& Facts, Color, Discovery. To make your life (and citation) easier just copy & paste the information below into your assignment or essay: That gives credibility to your paper and it is sometimes required in higher education. CitationWhen you need to include a fact or piece of information in an assignment or essay you should also include where and how you found that piece of information. How about an incentive to share this post? (You will help other colleagues find this blog)ĭownload and enjoy this complete and colored periodic table for you to edit and enjoy. Need an editable periodic table to edit? Maybe add your school logo, work team or anything else to maker your paper look cool?Īlong with basic atom / element information (like Potassium valence electrons and all the other atomic data), it also comes with color coded info about: State (Gas, Liquid or Solid at room temperature), Groups/series details and much more. How a small number of atoms can be joined and form completely different substances.Video Are you having trouble understanding the basics of atomic elements? This video will walk you through: Want to learn more details and data about Potassium (K)? Check my Elements Comprehensive List. Group 2 atoms lose two electrons to form. Pure metal is produced by the reaction of hot potassium chloride and sodium vapors in a special retort.ĭiscoveryDiscovered By: Sir Humphrey Davy For example, potassium atoms do this to form ions with the same electron configuration as the noble gas argon. 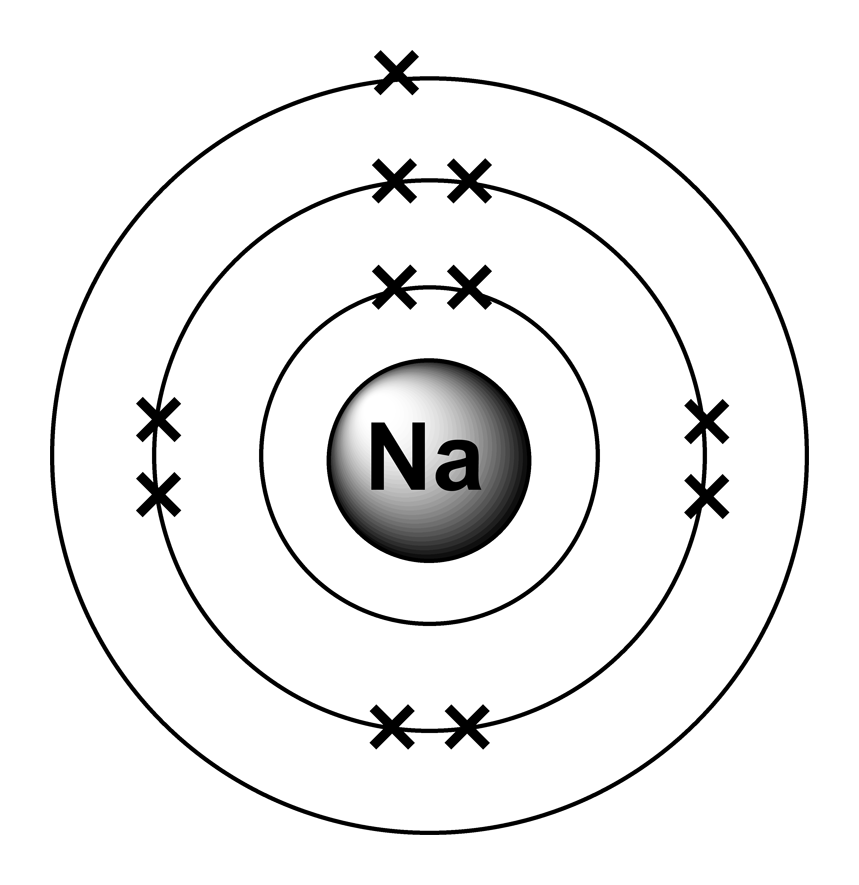
SourcesFound in minerals like carnallite & sylvite (potassium chloride, KCL). 
Name OriginEnglish: pot ash symbol from Latin: kalium, (alkali). Vital to function of nerve and muscle tissures. Also as saltpeter, potassium nitrate (KNO3) to make explosives and to color fireworks in mauve. UsesUsed as potash in making glass & soap. Eighth most abundant element in the earth's crust (20,900 ppm). In the case of Potassium the valence electrons is 1. Now let's check the facts about Potassium.ĭescriptionSoft, waxy, silver-white metal. Ok but how many valence electrons does an atom of Potassium have? 
A valence electron is an outer shell electron and may participate in the formation of a chemical bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
